
Body size mediates trophic interaction strength of novel fish assemblages under climate change
This blog post is provided by Minami Sasaki, Kelsey M. Kingsbury, David J Booth and Ivan Nagelkerken and tells the #StoryBehindthePaper for the paper “Body size mediates trophic interaction strength of… Continue reading Body size mediates trophic interaction strength of novel fish assemblages under climate change
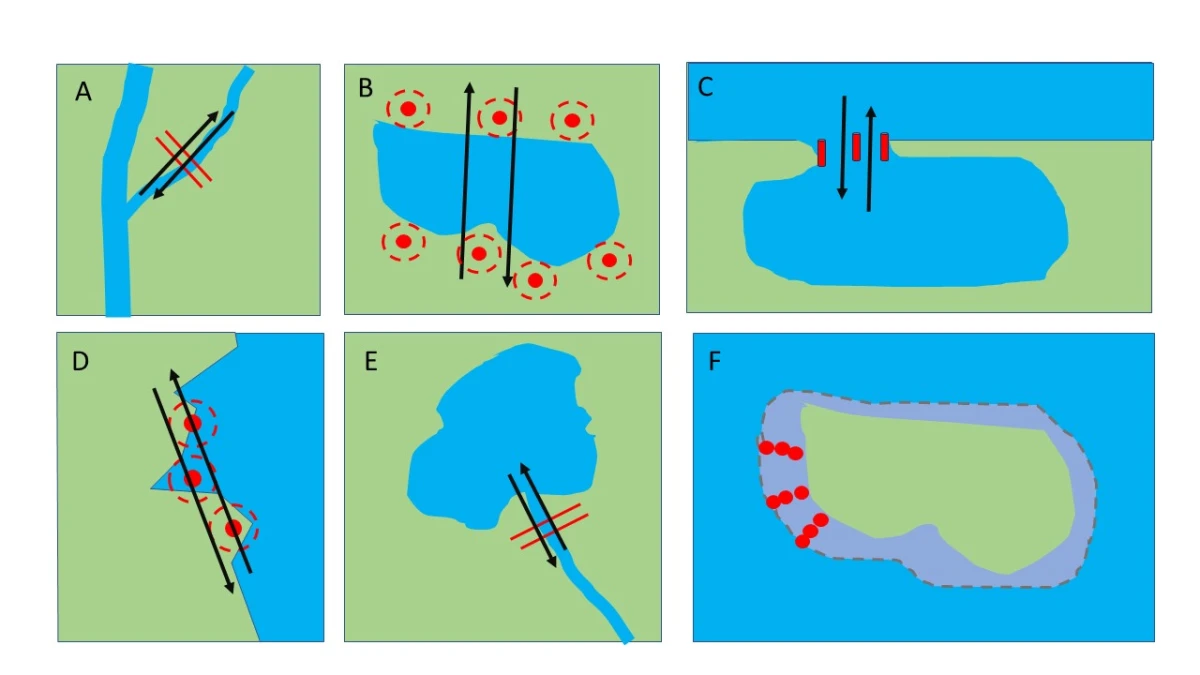
Estimating migration timing and abundance in partial migratory systems using automated telemetry
This blog post is provided by M. C. Dzul, W.L. Kendall, C.B. Yackulic, D.R. Van Haverbeke, P. Mackinnon, K. Young, M.J. Pillow & J. Thomas and tells the #StoryBehindThePaper… Continue reading Estimating migration timing and abundance in partial migratory systems using automated telemetry
Winner announced: 2023 Elton Prize
We are delighted to announce Clara Woodie as the 2023 winner of our Elton Prize early career researcher award Clara received the award for her paper, “long transients and… Continue reading Winner announced: 2023 Elton Prize

Modelling trampling effects of large herbivores on ecosystem processes
This blog post is provided by Adam Meyer and Shawn Leroux and tells the #StoryBehindthePaper for the paper “A theory for context-dependent effects of mammalian trampling on ecosystem nitrogen… Continue reading Modelling trampling effects of large herbivores on ecosystem processes

Garben Logghe – Our Elton Prize Shortlist Candidate’s #TheStoryBehindThePaper
Garben Logghe gives us information on his paper, “arthropod food webs predicted from body size ratios are improved by incorporating prey defensive properties“, which was shortlisted for this year’s… Continue reading Garben Logghe – Our Elton Prize Shortlist Candidate’s #TheStoryBehindThePaper

What kind of birds put artificial materials into their nests?
This blog post is provided by Catherine Sheard and tells the #StoryBehindThePaper for the paper “Anthropogenic nest material use in a global sample of birds”, which was recently published… Continue reading What kind of birds put artificial materials into their nests?

Förvaltning av vilda djurpopulationer i en föränderlig värld: fallet med Östersjöns gråsälar
Denna blog är skriven av Daire Carroll från Instutitionen för Biologi och Miljövetenskaper vid Göteborgs Universitet, här beskrivs #HistorienBakomArtikeln för den vetenskapliga artikeln “120-years of ecological monitoring data shows… Continue reading Förvaltning av vilda djurpopulationer i en föränderlig värld: fallet med Östersjöns gråsälar

Managing wildlife in a changing environment: the case of the Baltic grey seal
This blog post is provided by Daire Carroll from the Department of Biological and Environmental Science, Gothenburg University and tells the #StoryBehindthePaper for the paper “120-years of ecological monitoring… Continue reading Managing wildlife in a changing environment: the case of the Baltic grey seal

Joseph Woodman – Our Elton Prize Shortlist Candidate’s #TheStoryBehindThePaper
Joe Woodman gives us information on his paper, “disentangling the causes of age-assortative mating in bird populations with contrasting life-history strategies”, which was shortlisted for this year’s Elton Prize.… Continue reading Joseph Woodman – Our Elton Prize Shortlist Candidate’s #TheStoryBehindThePaper

Voles in space: food resources and intestinal parasites impact spatial behaviour in wild rodents
This blog post is provided by Janine Mistrick and tells the #StoryBehindThePaper for the paper “Effects of food supplementation and helminth removal on space use and spatial overlap in… Continue reading Voles in space: food resources and intestinal parasites impact spatial behaviour in wild rodents
Loading…
Something went wrong. Please refresh the page and/or try again.


